|
12/14/2022 0 Comments Mass number of krypton 
The standard SI unit is kilograms per cubic meter ( kg/m 3). In other words, the density (ρ) of a substance is the total mass (m) of that substance divided by the total volume (V) occupied by that substance. It is an intensive property, which is mathematically defined as mass divided by volume: Typical densities of various substances at atmospheric pressure.ĭensity is defined as the mass per unit volume. How does the atomic mass determine the density of materials? Density of Krypton The atomic mass number determines especially the atomic mass of atoms. The mass number is different for each different isotope of a chemical element. For 63Cu, the atomic mass is less than 63, so this must be the dominant factor. A nucleus with greater binding energy has lower total energy, and therefore a lower mass according to Einstein’s mass-energy equivalence relation E = mc 2. The nuclear binding energy varies between nuclei.This increases the mass of nuclei with more neutrons than protons relative to the atomic mass unit scale based on 12C with equal numbers of protons and neutrons. The neutron is slightly heavier than the proton.There are two reasons for the difference between mass number and isotopic mass, known as the mass defect: For example, 63Cu (29 protons and 34 neutrons) has a mass number of 63, and an isotopic mass in its nuclear ground state is 62.91367 u. For other isotopes, the isotopic mass usually differs and is usually within 0.1 u of the mass number. One unified atomic mass unit is approximately the mass of one nucleon (either a single proton or neutron) and is numerically equivalent to 1 g/mol.įor 12C, the atomic mass is exactly 12u, since the atomic mass unit is defined from it. One atomic mass unit is equal to 1.66 x 10 -24 grams. The unit of measure for mass is the atomic mass unit (amu). Therefore this resulting atomic mass is calculated from naturally-occurring isotopes and their abundance. Note that each element may contain more isotopes. 
How does the atomic number determine the chemical behavior of atoms? Atomic Mass of Krypton Since the number of electrons is responsible for the chemical behavior of atoms, the atomic number identifies the various chemical elements. Krypton light has many spectral traces, and krypton plasma is beneficial in shiny, high-powered fuel lasers (krypton ion and excimer lasers), each of which resonates and amplifies a unique spectral line.Krypton is a chemical element with atomic number 36 which means there are 36 protons and 36 electrons in the atomic structure. 
Krypton, like the different noble gases, is utilized in lighting and pictures. It is one of the rare gases which is used in fluorescent lamps. It also occurs in traces in the atmosphere. It is colourless, odourless and tasteless noble gas. With rare exceptions, krypton is chemically inert. It is a colourless, odourless, tasteless noble gasoline that is found rarely in the environment and is often used with some rare gases in fluorescent lamps. Krypton is an element which is denoted by Kr and atomic number 36. Since protons and neutrons have about the same mass (and the mass of the electrons is negligible for lots purposes) and the mass defect of nucleon binding is continually small as compared to the nucleon mass, the atomic mass of any atom, while expressed in unified atomic mass devices (making a quantity called the "relative isotopic mass"), is within 1% of the complete wide variety A. The sum of the atomic number Z and the number of neutrons N present gives the mass number A of an atom. In an uncharged atom, the proton number or atomic number is likewise equal to the number of electrons present. 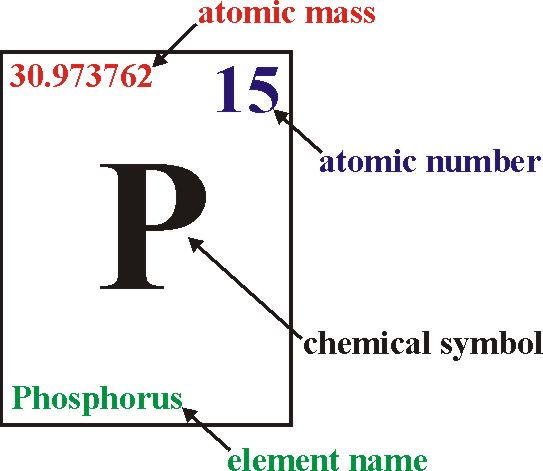
It is identical to the number of charges present in the nucleus. The atomic number uniquely identifies a chemical element. The atomic number (symbol Z) of an element is the number of protons found within the nucleus of each atom of that detail. The atomic number is written at the left hand side at top of each element. The atomic number is also equal to the number of electrons. The number of protons is equal to the atomic number. Hint:Atomic number is the number of the chemical compound which is present in the periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
